By Harrison Tasoff, UCSB Current
Scientists have solved a cellular murder mystery nearly 25 years after the case went cold. Following a trail of evidence from fruit flies to mice to humans revealed that cannibalistic cells likely cause a rare human immunodeficiency. Now the discovery shows promise for enhancing an up-and-coming cancer treatment.
“This paper takes us from very fundamental cell biology in a fly, to explaining a human disease and harnessing that knowledge for a cancer therapy,” said UC Santa Barbara’s Denise Montell. “Each one of those steps feels like a major discovery, but here they are, all in one paper.”
Researchers in Montell’s lab published their findings in the Proceedings of the National Academy of Sciences and are now investigating the mechanisms and implications.
An ancient gene
The primary character in this story is a gene, Rac2, and the protein it encodes. Rac2 is one of three Rac genes in humans. “Rac is very ancient in evolution, so it must serve a fundamental function,” said senior author Montell, Duggan Professor and Distinguished Professor of Molecular, Cellular, and Developmental Biology.
Rac proteins help build a cell’s scaffolding, called the cytoskeleton. The cytoskeleton is made of dynamic filaments that allow cells to maintain their shape or deform, as needed. In 1996, while studying a small group of cells in the fruit fly ovary, Montell determined that Rac proteins are instrumental in cell movement. Since then, it has become clear that Rac is a nearly universal regulator of cell motility in animal cells.
Back in the ‘90s, she noticed that a hyperactive form of the Rac1 protein, expressed in only a few cells in a fly’s egg chamber, destroyed the whole tissue. “Just expressing this active Rac in six to eight cells kills the entire tissue, which is composed of about 900 cells,” explained lead author Abhinava Mishra, a project scientist in Montell’s lab.
Why did this happen? How does it work? “This was our 25-year-old cold case,” Montell said.
A few years ago, evidence began to mount implicating cell eating, also known as cannibalism, in tissue destruction. There’s a step in normal fly egg development where certain cells similar to the border cells consume their neighbors because they are no longer needed. Indeed, cellular cannibalism is not as rare as you might expect: Millions of old red blood cells are eliminated from the human body this way every second.
Rac2 is one component of the complex eating process. Rac helps the eating cell to envelop its target. The team was curious if a hyperactive form of the protein was causing border cells to prematurely consume their neighbors.
For this to occur, the border cells need to recognize their targets, which requires a particular receptor. Indeed, when Mishra blocked this receptor, the border cells expressing activated Rac didn’t consume their neighbors, and the egg chamber remained alive and healthy.
“Our 25-year-old cold case was solved, and that was very satisfying for us,” Montell exclaimed. “But this is a fairly niche area of Drosophila egg development.” The implications would soon grow, though.
A mysterious immune condition
Around the time that her lab made their breakthrough, Montell caught wind of an intriguing study in the journal Blood. This paper found that three unrelated people suffering from recurrent infections had the exact same mutation, which hyperactivates Rac2, a Rac protein produced in blood cells. She suspected her lab’s recent revelation in fruit flies might shed light on this enigma.
The patients’ mutation was just mildly activating, and yet it was enough that they all suffered from multiple infections and ultimately needed bone marrow transplants. Blood tests revealed that these patients had nearly no T cells, a specialized kind of white blood cells crucial to the immune system. The team at the National Institutes of Health inserted the Rac2 mutation into mice and found the same mysterious loss of T cells. They also found that the T cells with hyperactive Rac developed normally in the animals’ bone marrow, and migrated to the thymus, where they continued to mature without incident. But then they just seemed to disappear. So, the paper ended with a mystery: what was causing the T cells to disappear?
The authors of that journal study had noticed that many of the patients’ neutrophils — another type of white blood cell — were enlarged. They seemed to be consuming quite a lot of material, unusual behavior in an otherwise healthy person.
Montell wondered if the patients’ T cells were disappearing because their innate immune cells like neutrophils with active Rac2 were eating them, much like the fruit fly border cells with active Rac were eating the egg chamber. Her team turned their attention to macrophages — the neutrophil’s more voracious counterpart — to investigate. Mishra cultured human macrophages with and without hyperactive Rac2 together with T cells. He observed that macrophages with hyperactive Rac consumed more cells, confirming the group’s hypothesis from their work with fruit flies.
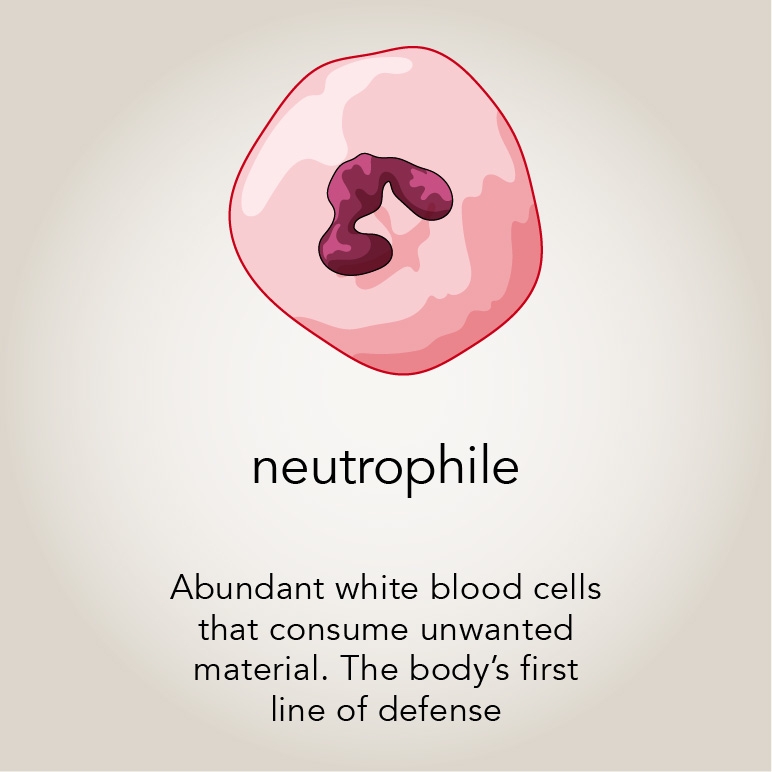
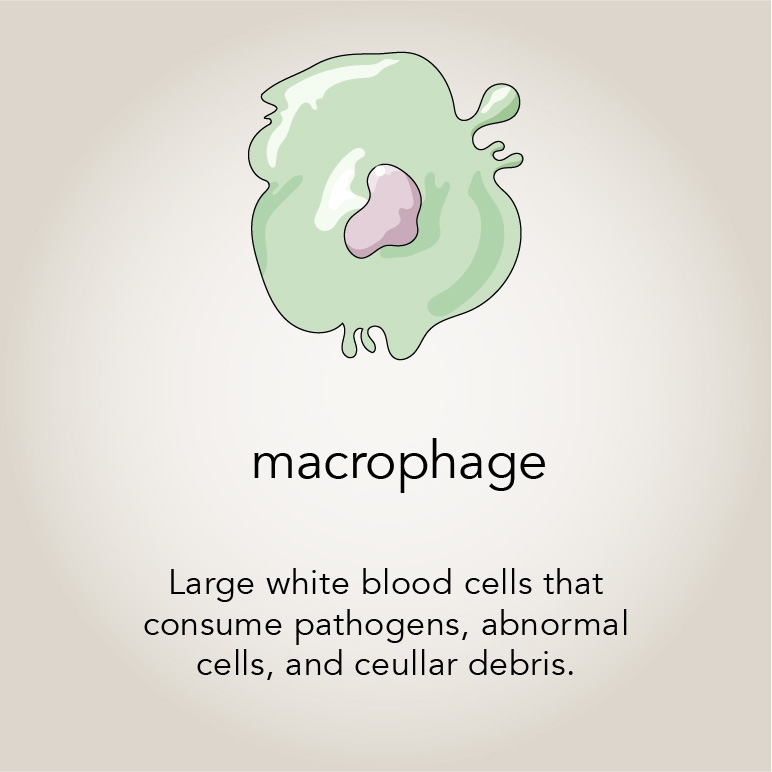

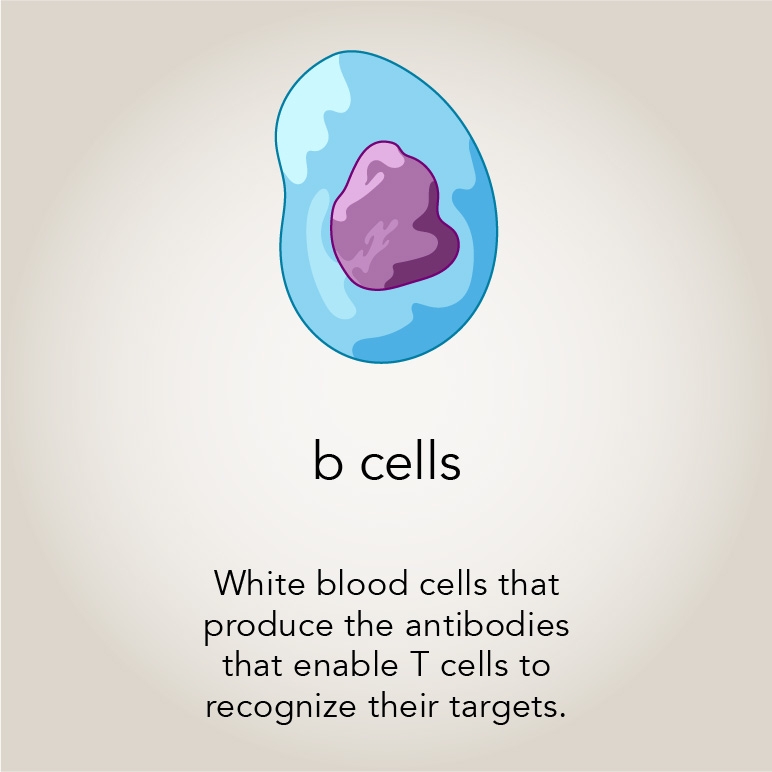
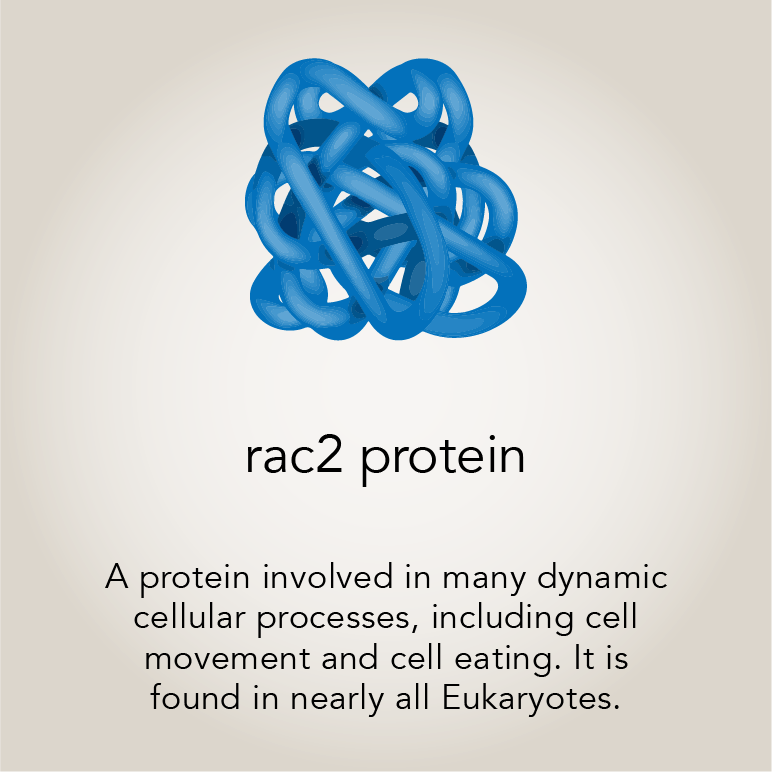

Photo Credit: Matt Perko
To test whether this might cause the observed immunodeficiency, co-author Melanie Rodriguez (a graduate student in Montell’s lab) took bone marrow samples from mice with the same hyperactive Rac2 mutation found in the patients. She then grew the marrow stem cells into macrophages, and performed a similar experiment to Mishra, but this time mixing both macrophages and T cells with and without the Rac2 mutation.
She found that macrophages with active Rac2 consumed significantly more T-cells than their normal counterparts. However, T-cells with active Rac2 were also more vulnerable to consumption from either kind of macrophage. So the most likely explanation for the patients’ missing T cells was a combination of increased consumption by macrophages as well as increased vulnerability of the T cells themselves. A human medical mystery was solved based on fundamental observations in fruit flies.
Harnessing haywire cells
The implications of these insights expanded in January 2020, when co-author Meghan Morrissey interviewed for a faculty position at UCSB. In her talk she described programming macrophages to eat cancer cells as a novel treatment for the disease, an approach called CAR-M. Morrissey had found that adding a CAR receptor to macrophages promoted this behavior. But it was also clear that inducing the macrophages to eat more would make the approach more effective — especially if they would specifically consume, and kill, entire cancer cells .
Well, if there was one thing that Montell and her lab had learned, it was how to make macrophages eat and kill whole, living cells. So they collaborated with Morrissey, now an assistant professor of molecular, cellular and developmental biology, to determine if adding activated Rac2 would increase the effectiveness of the CAR-M approach.
Rodriguez grew macrophages from the bone marrow of normal and mutant mice with activated Rac2. In each of these groups, Morrissey expressed either a dummy receptor or the CAR receptor, which recognizes B cells (another type of white blood cell). They found that the normal and hyperactive Rac cells with the dummy receptors did not eat many B cell targets. The normal macrophages with CAR receptors consumed far more B cells, as Morrissey had previously shown. However the macrophages with both hyperactive Rac and the CAR receptors ate twice-again as many B cells as the CAR-only group. Activated Rac2 also seemed to increase the number of so-called “super eaters” — ravenous macrophages that eat and kill multiple cancer cells.
The results made it clear that activated Rac and the receptor were both necessary for the enhanced effect. “If you add active Rac without the right receptor, it doesn’t do anything,” Montell explained.
This level of control is good news for any potential treatments, because it would give doctors a way to focus the modified macrophages’ attack on cancerous cells. Clinicians hopefully won’t need to worry about the engineered cells eating the patient’s T-cells either, because the T-cells wouldn’t have the active Rac2 mutation making them more vulnerable to this, as Rodriguez had previously discovered.
There is a current cancer treatment called CAR-T, which uses the CAR receptor and a patient’s own T-cells to attack and destroy cancers. It is highly effective against some cancers, but there are many that do not respond. CAR-M,a newer cousin to CAR-T, has recently entered into clinical trials in humans and so far seems safe. Montell and her group are interested in harnessing Rac-enhanced CAR macrophages to increase the efficacy of CAR-M treatments. They’ve filed a provisional patent for the technique — which they call RaceCAR-M — and are inviting biotech companies to partner in further developing the approach.

Further down the line, Montell wants to know how many kinds of cancer the RaceCAR-M treatment might successfully target. For comparison, CAR-T has been effective against cancers like leukemia and lymphoma, but not against solid-tumor cancers like breast, lung or colon.
The results have amazed Montell, an esteemed cell biologist with well over 100 papers to her name. “This is my favorite paper so far,” she said.
“We had this 25-year-old cold case in fruit flies, and we solved it,” Montell added. “And that helped us solve the mystery of an unexplained human immunodeficiency. And then we harnessed that knowledge to enhance a potential cancer immunotherapy.
“It was just one mystery after another, and Rac turned out to be the answer to each of them.”
You can hear more about the path from fruit flies to potential cancer treatment in Montell’s SNPets interview with the Genetics Society of America, episodes 4-6.






